
Continuing with this weeks Easter theme, the boys and I did some fun egg-periments. We did 5 experiments in total, however, today we are only going to talk about 3 cool experiments using 1 egg. Look for the other experiments ove the next few days.
The experiments today all evolve around making a rubber egg. Yep, you read that we made a rubber egg! Full disclosure, this experiment isn’t a fast one. It will take place over several days. Though there isn’t an immediate result, the cool part is that it gives you plenty of time to talk through the actual scientific process of what’s going go-which is a lot to process, especially for younger children.
Ingredients & Instructions

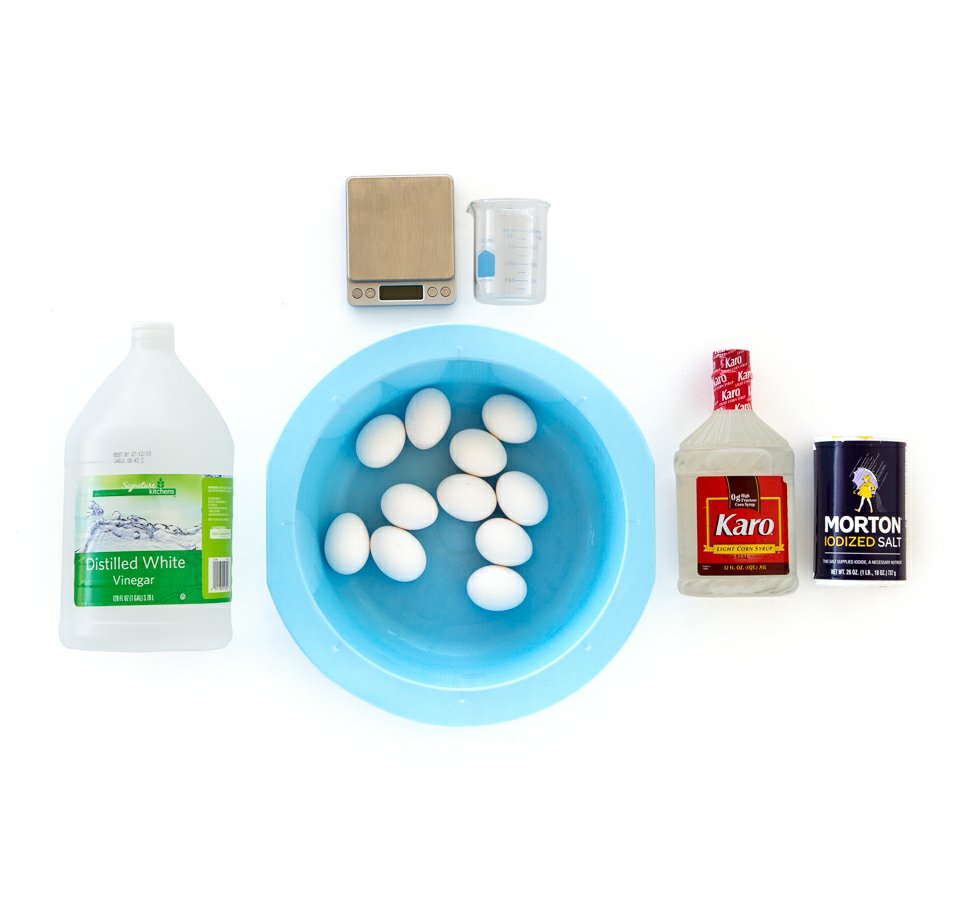
Steps:
- Get a jar, cup, or bowl and place an egg in it. I recommend making several jars, just in case one breaks. Also, this is a great way to try different acidic liquids at once, such as soda, orange juice, or coffee.
- Place the egg in vinegar solution.
- Wait 24 hrs and check egg.
- I found that it takes a little longer than 24 hrs to fully dissolve the shell. You can add fresh vinegar to the solution at this time to make the process faster.
- After 36 hrs, the egg shell has dissolved. Take the egg and gently rinse off the shell residue.
- Have your children observe the egg changes. Let them gently prod it and bounce it. Compare it to an unaltered egg to show them how much it has grown. I recommend doing this over a cookie sheet in case the egg breaks.
- After experimenting with the rubber egg, gently place it into corn syrup.
- Leave in corn syrup for 24 hrs.
- After 24 hrs, have your child observe the changes. The egg will have significantly shrunk at this point. The egg yolk will also feel kind of hard.
- After experimenting with the shrunken egg, gently place it into a glass of water. For added effects, add food coloring at this stage.
- After 24 hrs take it out and show it to your children. The egg has been rehydrated and the water has been reabsorbed into the membrane.
- At this point, have them experiment with the egg. Then either drop the egg from very high or pop the membrane. You can also place a pin or thumbtack into the outside of the membrane and show them how water squirts out.
- Once the egg is popped have them look at the yolk. Show them how the food color was absorbed through the membrane and changed the yolks color.

Egg Science
Eggshells contain calcium carbonate, which reacts with an acid in vinegar called acetic acid. The acetic acid breaks up the calcium and carbonate, dissolving the shell. While the calcium floats away, the carbonate reacts with the acetic acid to create carbon dioxide. This is why you see bubbles on and around the eggs.
To demonstrate this for smaller children, take their shirt sleeve and blow through it. Ask them if they felt the air through the shirt. Explain that their shirt sleeve is like the eggs membrane. Both has small holes that allow things to pass through them.
Osmosis, is the movement of a liquid, like water, across a membrane. Explain that membranes like to be balanced on both sides. The vinegar solution is mostly water with only a little vinegar in it, while inside the membrane is protein with a little water. So the water molecules travel from the vinegar into the egg to try to balance the concentrations, and the egg expands.
Continuing with the osmosis process, when they place the egg in corn syrup, it shrinks due to corn syrup being very sugary with only a little water dissolved in it. The egg will shrink because the membrane likes to stay balanced on both sides and will move water to do so.
Again with osmosis, when you place the shriveled egg in water, the water absorbs through the membrane to stay balanced. If you add food coloring, when you pop the egg, they will visually be able to see how the colored water was absorbed.
Further Investigation/For Older Children
Here are some steps for your more advanced scientists:
- Ask yourself questions What other acidic liquids would dissolve an eggshell? Would soda, tomato juice, or citrus juice work? Why does the color seep through the membrane but the albumen doesn’t leak out of the membrane? Are some eggshells harder to dissolve than others?
- Research Do research online and at the library to try and predict the answer to your question. Read about how the layers of an egg work and how liquids pass through the membrane. Research other liquids that calcium carbonate might react with. Research other substances that calcium carbonate is found in. Is it found in bones?
- Make a hypothesis A hypothesis is your prediction of the answer to your question based on your research. It may or may not be true.
- Experiment! Test your hypothesis by testing the variables and documenting them. Be sure to take notes of each experiment and what happens; this is called your data.
- Draw a conclusion Based on your experiments form a conclusion. Was your hypothesis correct?
- Share your findings Create a presentation with your findings. Include your research, hypothesis, the data you collected, and your conclusions. Be sure to include images and samples!
Did you do this experiment? Tell us your results below


You’re doing such a great job baby girl! Love you!!!
????
Thanks for sharing
Thumbs up! Your blog is actually a pleasant piece of content. Thumbs up! I am really intrigued about how this might affect things in the long term but it’s probably hard to see them with all the current changes we see happening.
I love what you guys tend to be up too. This kind of clever work and exposure! Keep up the great works guys I’ve added you guys to my personal blogroll.|
very easy to understand explanation. fits me perfectly. from now on I will be your fan
I’d like to thank you for the efforts you have put in writing this website. I am hoping to check out the same high-grade content from you later on as well. In fact, your creative writing abilities has encouraged me to get my very own site now ?
your writing really helped me I know what steps to take.
As I website possessor I believe the content matter here is rattling wonderful , appreciate it for your hard work. You should keep it up forever! Good Luck.